
You can:
| Name | Somatostatin receptor type 2 |
|---|---|
| Species | Mus musculus (Mouse) |
| Gene | Sstr2 |
| Synonym | somatotropin release-inhibiting factor receptor SRIF-1 SS-2-R SS2-R SS2R [ Show all ] |
| Disease | N/A for non-human GPCRs |
| Length | 369 |
| Amino acid sequence | MEMSSEQLNGSQVWVSSPFDLNGSLGPSNGSNQTEPYYDMTSNAVLTFIYFVVCVVGLCGNTLVIYVILRYAKMKTITNIYILNLAIADELFMLGLPFLAMQVALVHWPFGKAICRVVMTVDGINQFTSIFCLTVMSIDRYLAVVHPIKSAKWRRPRTAKMINVAVWCVSLLVILPIMIYAGLRSNQWGRSSCTINWPGESGAWYTGFIIYAFILGFLVPLTIICLCYLFIIIKVKSSGIRVGSSKRKKSEKKVTRMVSIVVAVFIFCWLPFYIFNVSSVSVAISPTPALKGMFDFVVILTYANSCANPILYAFLSDNFKKSFQNVLCLVKVSGTEDGERSDSKQDKSRLNETTETQRTLLNGDLQTSI |
| UniProt | P30875 |
| Protein Data Bank | N/A |
| GPCR-HGmod model | N/A |
| 3D structure model | No available structures or models |
| BioLiP | N/A |
| Therapeutic Target Database | N/A |
| ChEMBL | CHEMBL3207 |
| IUPHAR | 356 |
| DrugBank | N/A |
| Name | Vapreotide |
|---|---|
| Molecular formula | C57H70N12O9S2 |
| IUPAC name | 10-(4-aminobutyl)-N-[1-amino-3-(1H-indol-3-yl)-1-oxopropan-2-yl]-19-[(2-amino-3-phenylpropanoyl)amino]-16-[(4-hydroxyphenyl)methyl]-13-(1H-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-7-propan-2-yl-1,2-dithia-5,8,11,14,17-pentazacycloicosane-4-carboxamide |
| Molecular weight | 1131.38 |
| Hydrogen bond acceptor | 13 |
| Hydrogen bond donor | 13 |
| XlogP | 3.6 |
| Synonyms | L-Tryptophanamide, D-phenylalanyl-L-cysteinyl-L-tyrosyl-D-tryptophyl-L-lysyl-L-valyl-L-cysteinyl-, cyclic (2->7)-disulfide Vapreotide (USAN/INN) D-Phenylalanyl-L-cysteinyl-L-tyrosyl-D-tryptophyl-L-lysyl-L-valyl-L-cysteinyl-L-tryptophanamide cyclic (2-7)-disulfide D07DSQ NSC_71306 [ Show all ] |
| Inchi Key | SWXOGPJRIDTIRL-UHFFFAOYSA-N |
| Inchi ID | InChI=1S/C57H70N12O9S2/c1-32(2)49-57(78)68-48(55(76)64-44(50(60)71)26-35-28-61-41-16-8-6-14-38(35)41)31-80-79-30-47(67-51(72)40(59)24-33-12-4-3-5-13-33)56(77)65-45(25-34-19-21-37(70)22-20-34)53(74)66-46(27-36-29-62-42-17-9-7-15-39(36)42)54(75)63-43(52(73)69-49)18-10-11-23-58/h3-9,12-17,19-22,28-29,32,40,43-49,61-62,70H,10-11,18,23-27,30-31,58-59H2,1-2H3,(H2,60,71)(H,63,75)(H,64,76)(H,65,77)(H,66,74)(H,67,72)(H,68,78)(H,69,73) |
| PubChem CID | 71306 |
| ChEMBL | N/A |
| IUPHAR | N/A |
| BindingDB | 82464 |
| DrugBank | N/A |
Structure | 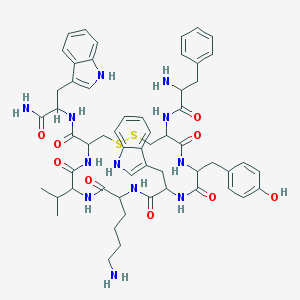 |
| Lipinski's druglikeness | This ligand has more than 5 hydrogen bond donor. This ligand has more than 10 hydrogen bond acceptor. This ligand is heavier than 500 daltons. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| Ki | 0.1 nM | PMID7870182 | BindingDB |
| Ki | 0.57 nM | PMID9600011 | BindingDB |
| Ki | 795.0 nM | PMID9600011 | BindingDB |
zhanglab![]() zhanggroup.org | +65-6601-1241 | Computing 1, 13 Computing Drive, Singapore 117417
zhanggroup.org | +65-6601-1241 | Computing 1, 13 Computing Drive, Singapore 117417