
You can:
| Name | Cannabinoid receptor 1 |
|---|---|
| Species | Homo sapiens (Human) |
| Gene | CNR1 |
| Synonym | CB1 Central cannabinoid receptor SKR6R THC receptor CB1R [ Show all ] |
| Disease | Obesity; Diabetes Chemotherapy-induced nausea Diabetes; Obesity Drug abuse Hypertension; Diabetes; Obesity [ Show all ] |
| Length | 472 |
| Amino acid sequence | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQEKMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIAVLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVFHRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLMWTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWKAHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLLAIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQPLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL |
| UniProt | P21554 |
| Protein Data Bank | 5tjv, 5u09, 5xr8, 5xra, 6n4b, 5tgz |
| GPCR-HGmod model | P21554 |
| 3D structure model | This structure is from PDB ID 5tjv. |
| BioLiP | BL0384680, BL0364157, BL0384679, BL0384681, BL0384682, BL0384683, BL0384684, BL0440253, BL0440254,BL0440255, BL0363267, BL0361447, BL0361446 |
| Therapeutic Target Database | T76685 |
| ChEMBL | CHEMBL218 |
| IUPHAR | 56 |
| DrugBank | BE0000061 |
| Name | Ibipinabant |
|---|---|
| Molecular formula | C23H20Cl2N4O2S |
| IUPAC name | (4S)-5-(4-chlorophenyl)-N-(4-chlorophenyl)sulfonyl-N'-methyl-4-phenyl-3,4-dihydropyrazole-2-carboximidamide |
| Molecular weight | 487.399 |
| Hydrogen bond acceptor | 4 |
| Hydrogen bond donor | 1 |
| XlogP | 5.3 |
| Synonyms | (4s)-(-)-3-(4-chlorophenyl)-N-methyl-N'-[(4-chlorophenyl)sulfonyl]-4-phenyl-4,5-dihydro-1h-pyrazole-1-carboxamidine 1794AH CHEMBL412262 Ibipinabant; SLV 319, (S)-(-)- SLV 319 [ Show all ] |
| Inchi Key | AXJQVVLKUYCICH-OAQYLSRUSA-N |
| Inchi ID | InChI=1S/C23H20Cl2N4O2S/c1-26-23(28-32(30,31)20-13-11-19(25)12-14-20)29-15-21(16-5-3-2-4-6-16)22(27-29)17-7-9-18(24)10-8-17/h2-14,21H,15H2,1H3,(H,26,28)/t21-/m1/s1 |
| PubChem CID | 9826744 |
| ChEMBL | CHEMBL412262 |
| IUPHAR | 9234 |
| BindingDB | 50230537 |
| DrugBank | N/A |
Structure | 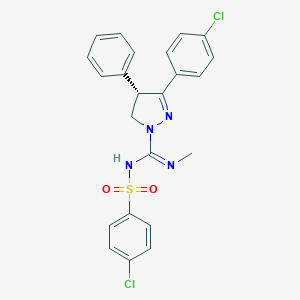 |
| Lipinski's druglikeness | This ligand has a partition coefficient log P greater than 5. |
| Parameter | Value | Reference | Database source |
|---|---|---|---|
| Activity | 4.09 pmol | PMID18207393 | ChEMBL |
| IC50 | 17.0 nM | PMID17181138 | ChEMBL |
| IC50 | 19.95 nM | PMID19338356 | ChEMBL |
| Kd | 0.1259 nM | PMID16140010, PMID20137935, PMID14736243 | ChEMBL |
| Ki | 0.42 nM | PMID21962575 | ChEMBL |
| Ki | 3.162 nM | PMID19338356 | ChEMBL |
| Ki | 4.1 nM | PMID18083560 | ChEMBL |
| Ki | 7.8 nM | PMID14736243 | IUPHAR |
| Ki | 7.8 nM | PMID16140010, PMID20137935, PMID18363352, PMID19520572, PMID14736243 | BindingDB,ChEMBL |
| Ki | 8.0 nM | PMID18335976 | ChEMBL |
zhanglab![]() zhanggroup.org | +65-6601-1241 | Computing 1, 13 Computing Drive, Singapore 117417
zhanggroup.org | +65-6601-1241 | Computing 1, 13 Computing Drive, Singapore 117417